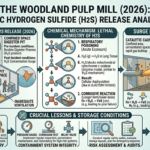
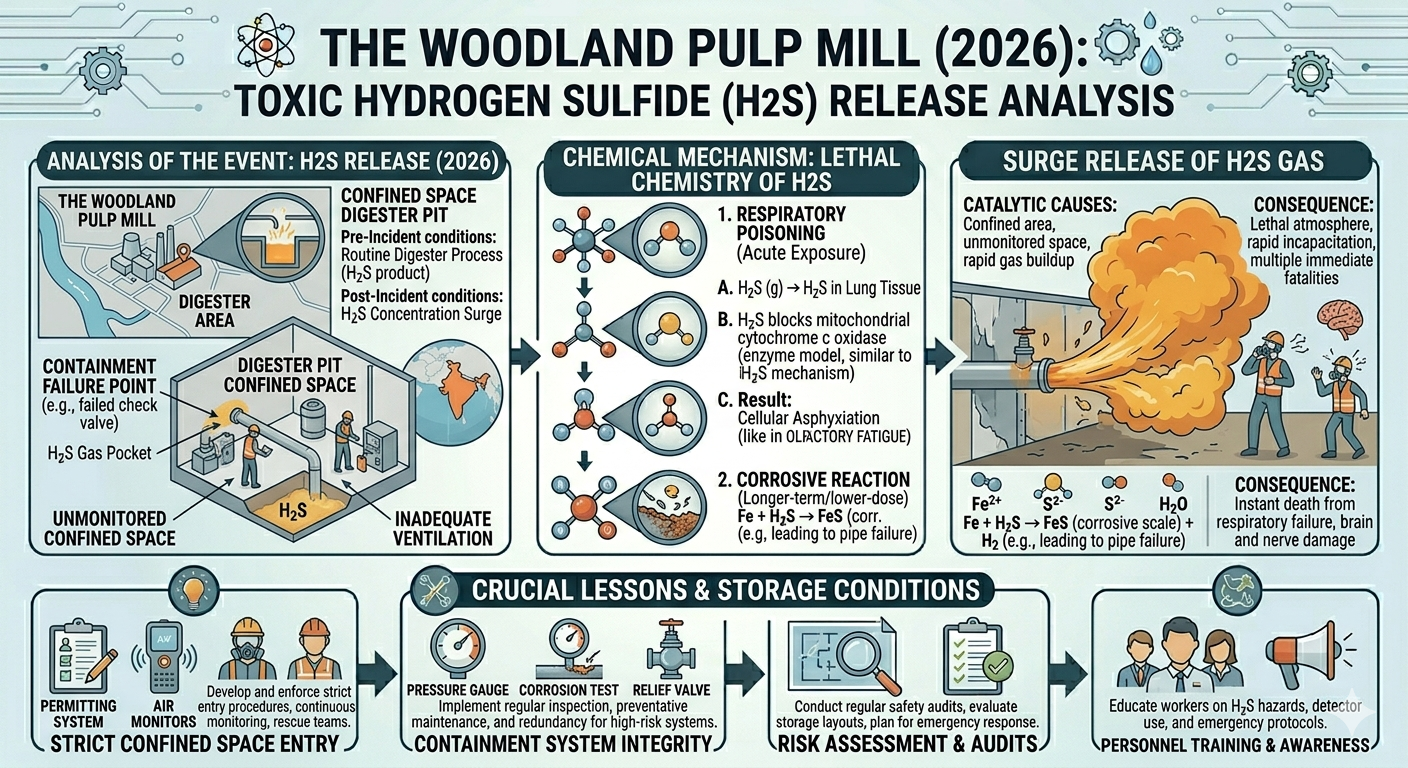
Industrial safety is often tested not by rare, unknown hazards, but by well-understood substances behaving exactly as chemistry predicts under unsafe conditions. The reported hydrogen sulfide (H₂S) release at the Woodland Pulp Mill in 2026 is a stark example of this reality. Hydrogen sulfide is a familiar industrial gas, especially in pulp and paper operations, yet it remains one of the most dangerous due to its toxicity, rapid action, and deceptive sensory properties. The incident underscores two critical themes: the failure of containment systems and the lethal chemistry of H₂S in confined spaces.
Understanding this event requires a closer look at how hydrogen sulfide behaves, how containment systems are designed to control it, and how multiple failures can align to create a life-threatening situation.
Hydrogen Sulfide: A Familiar but Deadly Gas
Hydrogen sulfide is a colorless gas with a characteristic “rotten egg” odor at low concentrations. It is commonly generated in industrial processes involving sulfur compounds, including petroleum refining, wastewater treatment, and pulp and paper manufacturing. In kraft pulping, which is widely used in the paper industry, sulfur-based chemicals are essential for breaking down lignin in wood, and H₂S can be produced as a byproduct.
Despite its common presence, hydrogen sulfide is extremely hazardous. Its toxicity is comparable to that of other well-known industrial poisons, and its effects on the human body are both rapid and severe. At low concentrations, it may only cause irritation, but at higher levels, it can lead to immediate collapse and death.
The Deceptive Nature of Detection
One of the most dangerous aspects of hydrogen sulfide is its effect on the human sense of smell. At very low concentrations, the gas is easily detected by its odor. However, as concentration increases, it quickly causes olfactory fatigue, a condition in which the sense of smell becomes desensitized. At this point, the gas is no longer detectable by odor, even though it may be present at lethal levels.
This creates a false sense of security. Workers may initially notice the smell and assume they can rely on it as a warning signal, only to lose that ability as concentrations rise. At dangerously high levels, hydrogen sulfide can incapacitate a person within seconds, leaving no time for escape.
The Chemistry of Toxicity
The lethality of hydrogen sulfide lies in its interaction with the body at a cellular level. It acts as a chemical asphyxiant, meaning it prevents cells from using oxygen effectively.
Inside the body, H₂S inhibits an enzyme known as cytochrome c oxidase, which plays a crucial role in the mitochondrial electron transport chain. This chain is responsible for producing energy in cells through aerobic respiration. When the enzyme is blocked, oxygen cannot be utilized, even if it is present in the bloodstream.
The result is a form of suffocation at the cellular level. Organs that require high amounts of oxygen, such as the brain and heart, are affected first. Symptoms can progress rapidly from dizziness and confusion to loss of consciousness, respiratory failure, and death.
At very high concentrations, this process can occur almost instantaneously, making hydrogen sulfide one of the most dangerous gases encountered in industry.
The Danger of Confined Spaces
While hydrogen sulfide is hazardous in any environment, it becomes especially dangerous in confined or poorly ventilated spaces. Several physical and chemical properties contribute to this increased risk.
First, hydrogen sulfide is slightly heavier than air. This means it tends to settle in low-lying areas such as pits, tanks, and trenches. In these locations, the gas can accumulate without being easily dispersed.
Second, confined spaces often lack adequate ventilation. Without sufficient airflow, any released gas remains trapped, allowing concentrations to rise quickly. Even a small leak can lead to dangerous conditions in a short period.
Third, the geometry of confined spaces can create pockets where gas accumulates unevenly. These “dead zones” may not be detected by sensors placed elsewhere, increasing the risk of unexpected exposure.
In such environments, a single release can create a lethal atmosphere almost instantly, particularly if workers are not equipped with proper protective equipment.
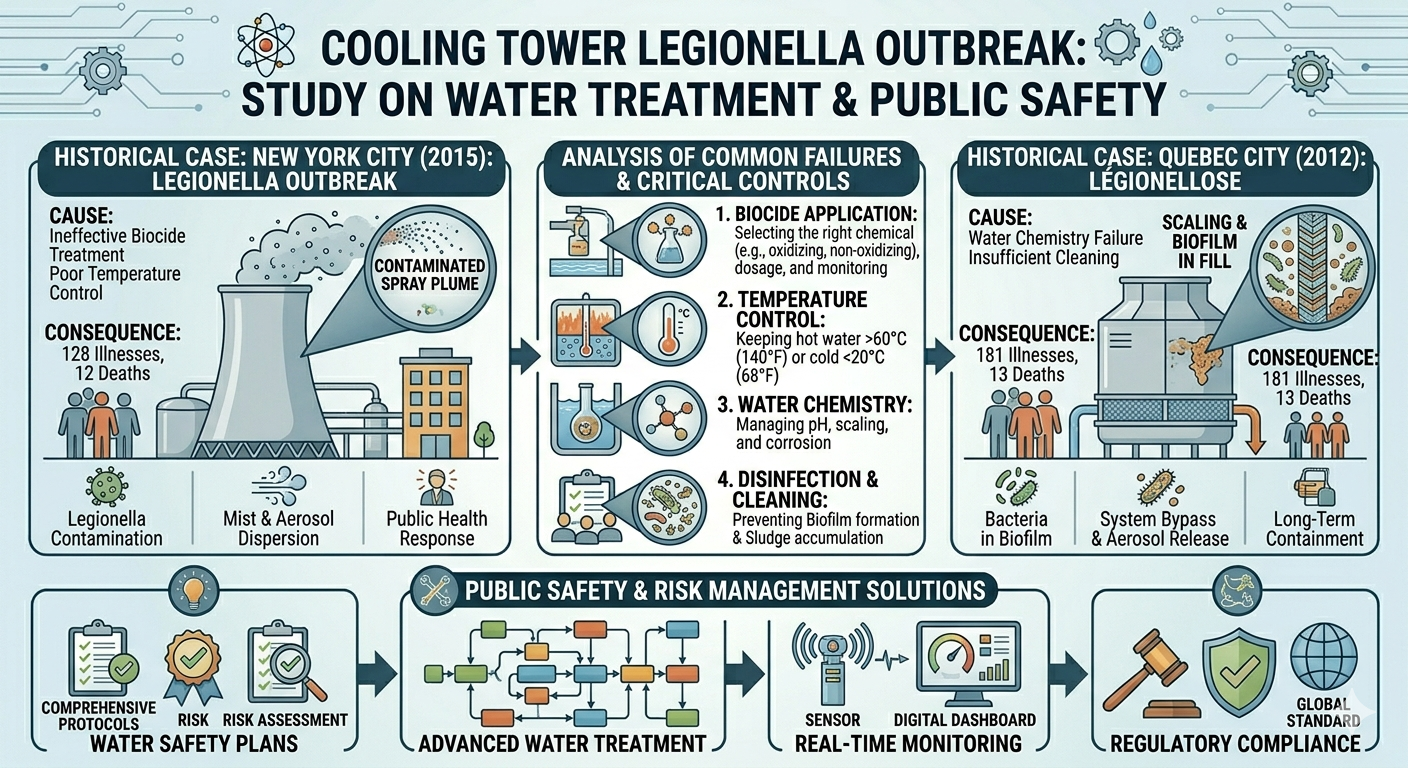
The Woodland Incident: Failures in Containment
Although full details of the Woodland Pulp Mill incident may still be under investigation, similar events typically involve a combination of failures rather than a single cause. Central to the issue is the breakdown of containment systems.
Containment systems in industrial facilities are designed to keep hazardous substances within controlled boundaries. These systems include pipelines, valves, seals, and pressure vessels, all engineered to handle specific operating conditions. When functioning correctly, they prevent the release of toxic gases like hydrogen sulfide.
However, several factors can compromise containment:
- Material degradation, such as corrosion or wear, can weaken pipes and seals
- Mechanical failure, including faulty valves or connections, can allow gas to escape
- Overpressure conditions can cause ruptures or leaks
- Improper maintenance can leave critical components in unsafe condition
In pulp mills, where processes often involve high temperatures and pressures, these risks are amplified. A small failure in a pressurized system can release significant amounts of gas in a very short time.
The Role of Detection and Monitoring Systems
Modern industrial facilities rely on gas detection systems to provide early warning of hazardous conditions. These systems include fixed sensors installed in key locations and portable monitors carried by workers.
For hydrogen sulfide, detection systems are particularly important due to the limitations of human senses. Properly functioning sensors can detect even low concentrations and trigger alarms before conditions become dangerous.
However, detection systems can fail if they are not properly maintained or calibrated. Sensors may drift over time, leading to inaccurate readings. Alarm thresholds may be set incorrectly, delaying response. In some cases, alarms may be ignored or misunderstood by personnel.
In the absence of reliable detection, workers may be exposed to dangerous concentrations without any warning.
Ventilation and Gas Dispersion
Ventilation plays a crucial role in controlling gas accumulation. In well-designed systems, airflow is used to dilute and remove hazardous gases, preventing them from reaching dangerous levels.
If ventilation systems fail or are inadequate, gases like hydrogen sulfide can accumulate rapidly. Blocked ducts, malfunctioning fans, or poor system design can all contribute to ineffective ventilation.
In confined spaces, the lack of ventilation is particularly dangerous. Without a mechanism to remove gas, concentrations can rise to lethal levels within seconds.
Human Factors and Emergency Response
Even with proper engineering controls, human factors play a significant role in industrial safety. Training, awareness, and adherence to procedures are essential for preventing and responding to incidents.
In hydrogen sulfide incidents, one of the most common and tragic patterns is the failure of rescue attempts. Workers who attempt to help affected colleagues without proper protective equipment often become victims themselves. This highlights the importance of strict protocols for confined space entry and emergency response.
Workers must be trained to recognize hazards, use detection equipment, and follow established procedures. Emergency response teams must be equipped with appropriate breathing apparatus and trained to operate in hazardous environments.
Prevention Strategies
Preventing hydrogen sulfide incidents requires a comprehensive approach that integrates engineering, monitoring, and human factors.
Key measures include:
- Robust containment systems, with regular inspection and maintenance
- Reliable gas detection, including calibration and testing of sensors
- Effective ventilation, designed to prevent gas accumulation
- Strict confined space procedures, including permits and pre-entry testing
- Comprehensive training, ensuring that workers understand risks and responses
- Emergency preparedness, with trained teams and proper equipment
These measures are not optional; they are essential components of safe operation in environments where hydrogen sulfide is present.
Conclusion
The Woodland Pulp Mill hydrogen sulfide release serves as a powerful reminder of the risks associated with toxic gases in industrial settings. Hydrogen sulfide is well understood, yet its dangers are often underestimated until it is too late.
From a chemical perspective, its ability to disrupt cellular respiration makes it a fast-acting and deadly hazard. From an engineering perspective, failures in containment, detection, and ventilation can allow this hazard to manifest with devastating consequences.
Ultimately, the lesson is clear: safety depends on respecting both the chemistry of hazardous substances and the systems designed to control them. Hydrogen sulfide does not provide second chances. In confined spaces, even a brief lapse in control can lead to irreversible outcomes.
By maintaining rigorous standards, investing in safety systems, and fostering a culture of vigilance, industries can prevent such incidents and protect the lives of those who work within them.