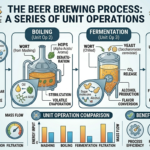
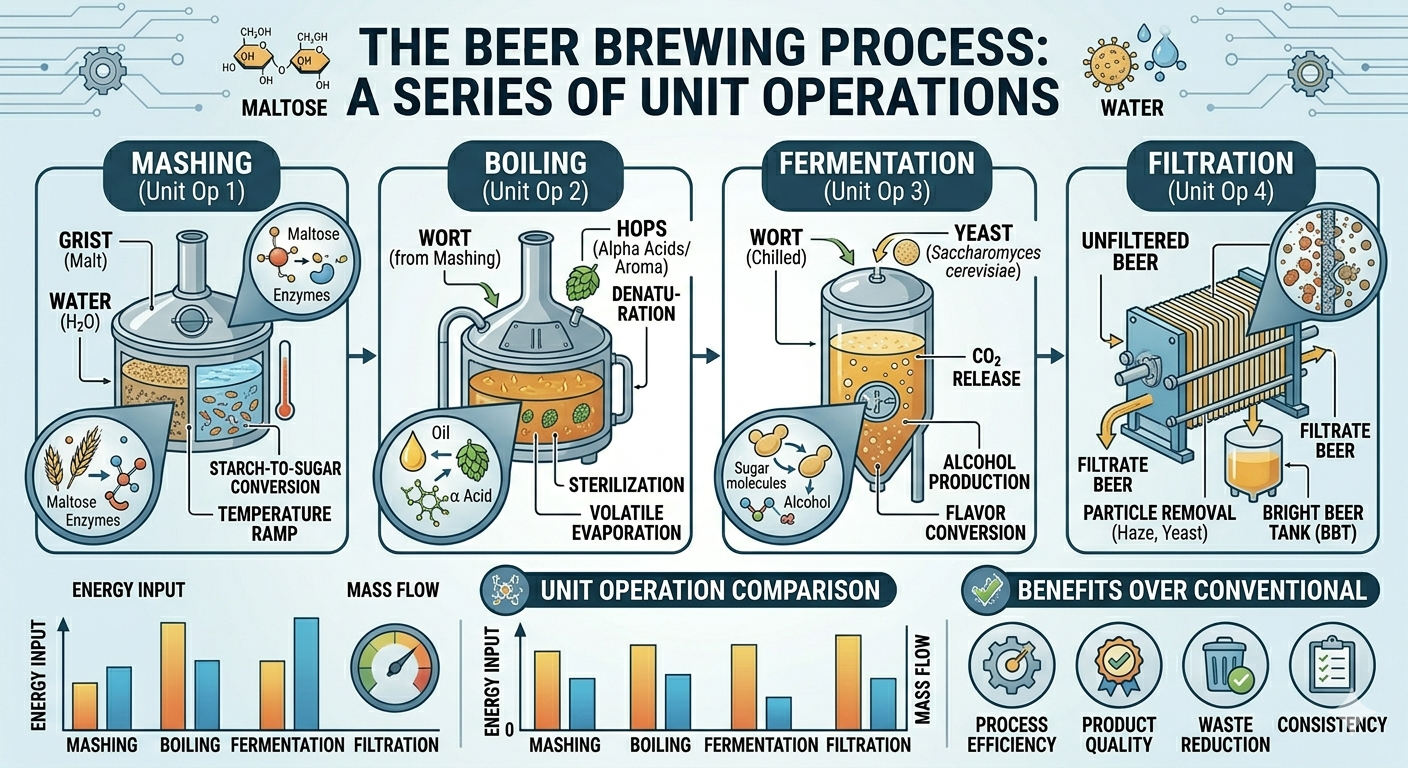
Beer is one of the oldest and most widely consumed beverages in the world, enjoyed across cultures for thousands of years. While it is often associated with craftsmanship, tradition, and creativity, the brewing process is fundamentally rooted in science—specifically, chemical engineering. Beneath the artistry of brewing lies a structured sequence of unit operations, the same building blocks used in large-scale industrial processes.
From transforming grains into sugars to fermenting those sugars into alcohol and finally clarifying the product, brewing follows a logical and efficient pathway. The four primary stages—mashing, boiling, fermentation, and filtration—mirror classical chemical engineering operations such as extraction, heat transfer, reaction engineering, and separation. Understanding beer brewing through this lens not only demystifies the process but also highlights its elegance as a well-designed system.
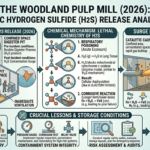
Mashing: Extraction and Enzymatic Conversion
The brewing process begins with mashing, where crushed malted grains, usually barley, are mixed with hot water to form a thick mixture known as mash. This stage is critical because it determines the composition of the liquid that will eventually become beer.
From a chemical engineering perspective, mashing involves two key unit operations: solid-liquid extraction and enzymatic reaction. The goal is to extract soluble compounds from the grains while converting complex starches into simpler, fermentable sugars.
Inside the mash, naturally occurring enzymes—primarily amylases—break down starch molecules into sugars such as maltose and glucose. These sugars serve as food for yeast in the later fermentation stage. The efficiency of this conversion depends heavily on temperature, making mashing a carefully controlled process.
Different temperature ranges activate different enzymes. Lower temperatures favor enzymes that produce highly fermentable sugars, resulting in a lighter, drier beer. Higher temperatures promote the formation of more complex sugars, contributing to a fuller body and richer mouthfeel. This is a clear example of reaction engineering, where process conditions are adjusted to control product outcomes.
In addition to temperature, factors such as pH, water composition, and mash time also influence extraction efficiency and sugar profile. Thus, mashing is not just a simple mixing step—it is a finely tuned operation that sets the foundation for the entire brewing process.
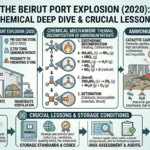
Boiling: Heat Transfer and Chemical Transformation
After mashing, the liquid portion, known as wort, is separated from the spent grains and transferred to a boiling vessel. The boiling stage serves multiple purposes, all of which are essential for producing a stable and flavorful beer.
From an engineering standpoint, boiling is primarily a heat transfer operation, where energy is supplied to raise the temperature of the wort to its boiling point and maintain it there for a specific duration. However, several important physical and chemical transformations occur during this stage.
First, boiling acts as a sterilization step, eliminating unwanted microorganisms that could spoil the beer. Second, it halts enzymatic activity from the mashing stage, effectively locking in the sugar composition of the wort.
One of the most important aspects of boiling is the addition of hops, which contribute bitterness, flavor, and aroma. During boiling, compounds in hops undergo chemical transformations that influence the final taste of the beer. This includes isomerization reactions that convert hop acids into more soluble and bitter forms.
Boiling also facilitates the removal of undesirable volatile compounds, such as dimethyl sulfide (DMS), which can produce off-flavors if not properly eliminated. Additionally, proteins in the wort coagulate and precipitate out, improving clarity and stability.
In essence, boiling is a combination of heat transfer, phase equilibrium, and reaction engineering, all working together to refine the wort before fermentation.
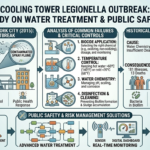
Fermentation: The Biochemical Reactor
Fermentation is the heart of the brewing process and the stage where beer truly comes to life. Once the wort is cooled to an appropriate temperature, yeast is added, initiating a complex series of biochemical reactions.
At its core, fermentation is a bioreactor operation, where living microorganisms convert sugars into alcohol and carbon dioxide. The primary reaction can be summarized as:
Glucose → Ethanol + Carbon Dioxide
This transformation is governed by principles of reaction kinetics, mass transfer, and microbial metabolism. Yeast consumes the sugars produced during mashing and releases ethanol, CO₂, and a variety of flavor compounds.
The conditions under which fermentation occurs have a significant impact on the final product. Temperature, in particular, plays a crucial role. Warmer fermentation temperatures typically result in faster reactions and the production of fruity or spicy flavors, characteristic of ales. Cooler temperatures lead to slower fermentation and cleaner, crisper profiles, typical of lagers.
Other important variables include oxygen levels, nutrient availability, and yeast strain. Each yeast strain has unique characteristics that influence flavor, aroma, and alcohol content. Managing these variables requires careful monitoring and control, much like in industrial fermentation processes used in pharmaceuticals or biotechnology.
Fermentation also produces heat, which must be removed to maintain optimal conditions. This introduces another layer of process control and heat management, reinforcing the engineering nature of brewing.
Filtration: Separation and Clarification
Once fermentation is complete, the beer contains not only alcohol and flavor compounds but also suspended particles such as yeast cells, proteins, and other solids. To improve clarity, stability, and appearance, these unwanted components must be removed.
This is where filtration and separation processes come into play. Depending on the desired style and production scale, brewers may use a combination of techniques, including filtration, centrifugation, and sedimentation.
Filtration involves passing the beer through a medium that traps solid particles while allowing the liquid to pass through. Centrifugation, commonly used in large breweries, accelerates the separation process by applying centrifugal force to remove solids more efficiently.
The goal of this stage is not only aesthetic but also practical. Removing suspended particles helps extend shelf life and ensures consistent flavor. However, some beer styles intentionally retain a degree of cloudiness, as it contributes to their character and mouthfeel.
From a chemical engineering perspective, this stage represents phase separation, where solid and liquid components are divided to achieve the desired product quality.
Additional Operations in Modern Brewing
While mashing, boiling, fermentation, and filtration form the core of the brewing process, modern breweries often include additional steps that further refine the product.
Conditioning or aging allows the beer to mature, during which flavors develop and unwanted byproducts are reduced. This step can be viewed as a controlled holding process where equilibrium is gradually achieved.
Carbonation involves dissolving carbon dioxide into the beer, either naturally through fermentation or artificially by injection. This is a classic example of gas-liquid mass transfer, where CO₂ is absorbed into the liquid under pressure.
Finally, packaging ensures that the beer is stored and transported without contamination or oxidation. This involves careful control of environmental conditions to preserve quality.
Brewing as a Complete Process System
When viewed as a whole, beer brewing resembles a compact chemical processing plant. Each stage is interconnected, and the output of one operation becomes the input for the next. Engineers often represent such systems using process flow diagrams, highlighting the sequence of operations and material flows.
The integration of these steps allows brewers to optimize efficiency, control quality, and scale production from small craft batches to large industrial volumes. Modern breweries rely on sensors, automation, and data analysis to maintain consistency and improve performance.
Conclusion
The brewing of beer is far more than a simple craft—it is a sophisticated application of chemical engineering principles. By understanding the process as a series of unit operations—mashing, boiling, fermentation, and filtration—we gain insight into how raw ingredients are transformed into a complex and enjoyable beverage.
Each stage involves fundamental engineering concepts, from mass transfer and reaction kinetics to heat transfer and phase separation. Together, they form a cohesive system that balances science and creativity.
Ultimately, beer brewing exemplifies how traditional practices can align seamlessly with modern engineering. It is a process where precision meets passion, and where every glass reflects a carefully orchestrated sequence of operations designed to deliver both quality and enjoyment.