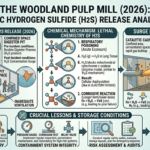
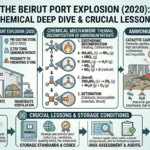
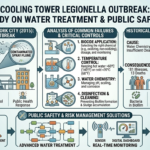
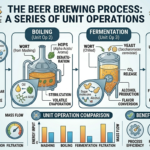
In recent decades, advances in science and engineering have steadily pushed the boundaries of what is possible in medicine. Among the most transformative of these innovations is microfluidics, a field that manipulates fluids at the microscale to perform complex biochemical processes. Often described as the foundation of “lab-on-a-chip” technology, microfluidics enables entire laboratory operations to be miniaturized onto a single chip no larger than a credit card. This revolutionary approach is reshaping diagnostics, drug development, and personalized medicine, offering faster, cheaper, and more accessible healthcare solutions.
Understanding Microfluidics
Microfluidics involves the precise control and manipulation of fluids in channels with dimensions typically ranging from a few micrometers to a few hundred micrometers. At this scale, fluid behavior differs significantly from what we observe in everyday life.
Re=ρvD/μ
The Reynolds number (Re) is a dimensionless parameter used to predict flow behavior. In microfluidic systems, the Reynolds number is usually very low, meaning that flow is laminar rather than turbulent. This has several important consequences. Fluids move in smooth, parallel streams without chaotic mixing, and mixing occurs primarily through diffusion rather than convection. While this might seem like a limitation, it actually provides a high degree of control, allowing engineers to design systems where reactions occur with remarkable precision.
The Lab-on-a-Chip Concept
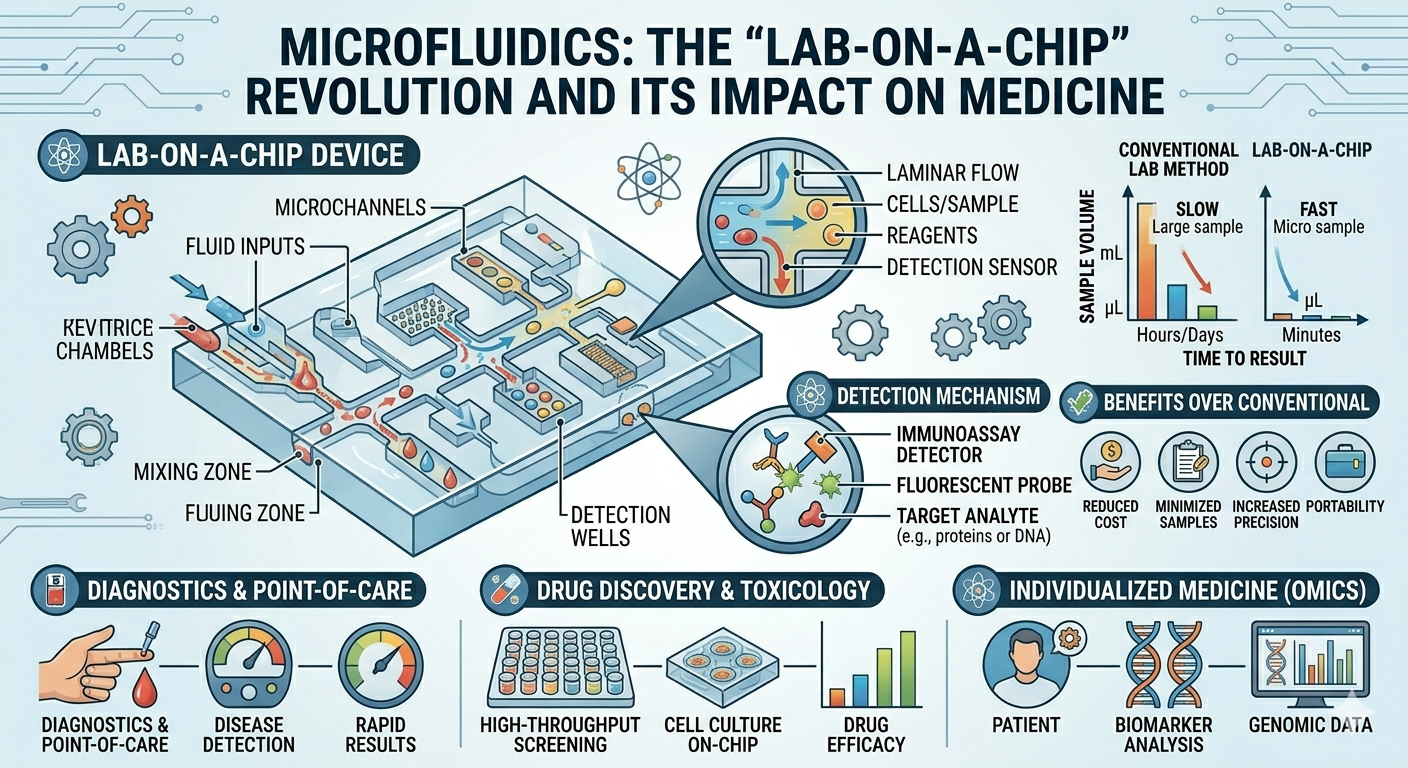
The concept of a lab-on-a-chip (LOC) is central to microfluidics. These devices integrate multiple laboratory functions—such as sample preparation, chemical reactions, separation, and detection—onto a single micro-scale platform. Instead of relying on bulky equipment and extensive manual handling, LOC systems automate and streamline processes, often delivering results within minutes.
For example, a small chip can analyze a drop of blood to detect biomarkers, identify pathogens, or measure glucose levels. This miniaturization reduces the need for large sample volumes and expensive reagents, making testing more efficient and cost-effective.
Transforming Medical Diagnostics
One of the most significant impacts of microfluidics is in the field of medical diagnostics, particularly in point-of-care testing. Traditional diagnostic methods typically require centralized laboratories, trained personnel, and long processing times. In contrast, microfluidic devices can provide rapid results at the patient’s bedside, in clinics, or even in remote locations.
This capability is especially valuable in situations where time is critical, such as detecting infectious diseases. Rapid diagnostic tests based on microfluidics have already been used for conditions like COVID-19, enabling faster identification and response. By reducing turnaround time, these devices improve patient outcomes and help control the spread of disease.
Enabling Personalized Medicine
Microfluidics is also playing a crucial role in the shift toward personalized medicine. By allowing high-throughput analysis of biological samples, microfluidic systems enable healthcare providers to tailor treatments to individual patients.
For instance, cancer treatment can be optimized by testing how a patient’s tumor cells respond to different drugs on a microfluidic chip. Similarly, genetic analysis can be performed quickly and efficiently, providing insights into a patient’s unique biological profile. This level of customization was previously difficult and expensive, but microfluidics is making it increasingly accessible.
Advancing Drug Development
The pharmaceutical industry is another area where microfluidics is having a profound impact. Drug development is traditionally a time-consuming and costly process, often requiring large-scale experiments and significant resources. Microfluidic systems offer a more efficient alternative.
By enabling precise control over reaction conditions and allowing multiple experiments to be conducted simultaneously, microfluidics accelerates the screening of drug candidates. Researchers can test various compounds, concentrations, and conditions on a single chip, reducing both time and cost.
Moreover, microfluidics supports the development of more accurate models for studying drug behavior. This leads to better predictions of how drugs will perform in real-world conditions, ultimately improving the success rate of new therapies.
Organ-on-a-Chip Technology
One of the most exciting applications of microfluidics is the development of organ-on-a-chip systems. These devices replicate the structure and function of human organs on a microscale, providing a more realistic environment for studying biological processes.
For example, a lung-on-a-chip can mimic the breathing motion and air–blood interface of human lungs, while a liver-on-a-chip can simulate metabolic processes. These systems allow researchers to study disease progression, test drug toxicity, and explore new treatments with greater accuracy than traditional cell cultures.
Organ-on-a-chip technology also has the potential to reduce reliance on animal testing, addressing ethical concerns and improving the relevance of experimental results to human health.
Chemical Engineering Principles Behind Microfluidics
Microfluidics is deeply rooted in chemical engineering, drawing on several key principles:
Fluid Dynamics
At the microscale, fluid behavior is dominated by viscous forces rather than inertial forces. Engineers must carefully design channel geometries to control flow rates and ensure consistent performance.
Mass Transport
Since mixing occurs primarily through diffusion, efficient transport of molecules is critical. Engineers use innovative designs, such as serpentine channels and micro-mixers, to enhance mixing and reaction rates.
Surface Chemistry
Surface effects become highly significant at small scales. By modifying surface properties, engineers can control how fluids interact with channel walls, enabling selective binding of molecules such as proteins or antibodies.
Materials and Fabrication
Microfluidic devices are typically made from materials like polydimethylsiloxane (PDMS), glass, or silicon. Fabrication techniques such as soft lithography allow precise construction of microchannels, enabling complex designs.
Advantages of Microfluidic Systems
Microfluidics offers several advantages that make it particularly well-suited for medical applications:
- Reduced sample and reagent consumption, lowering costs
- Rapid analysis, enabling quick decision-making
- High precision and reproducibility
- Portability, allowing use in remote or resource-limited settings
- Integration of multiple functions on a single device
These benefits contribute to more efficient and accessible healthcare.
Challenges and Limitations
Despite its potential, microfluidics faces several challenges that must be addressed for widespread adoption.
One major issue is scalability. While microfluidic devices are effective in laboratory settings, mass production can be complex and costly. Ensuring consistent quality across large numbers of devices remains a challenge.
Another limitation is integration with existing medical systems. Healthcare infrastructure is often built around traditional laboratory methods, and incorporating new technologies requires significant adaptation and validation.
Sensitivity is also a concern. Detecting extremely low concentrations of biomarkers may require advanced detection techniques, which can increase complexity and cost.
Finally, regulatory approval is a critical hurdle. Medical devices must meet strict standards for safety and reliability, which can slow the introduction of new technologies.
Real-World Applications
Microfluidics is already making a tangible impact in various areas of medicine. Examples include:
- Portable glucose monitoring devices for diabetes management
- Rapid diagnostic tests for infectious diseases
- DNA sequencing and genetic analysis tools
- Detection of cancer biomarkers
These applications demonstrate the versatility and effectiveness of microfluidic systems in real-world healthcare settings.
The Future of Microfluidics in Medicine
The future of microfluidics is promising, with ongoing research and innovation driving new developments. Emerging trends include the integration of microfluidics with artificial intelligence, enabling smarter and more automated diagnostic systems. Advances in 3D printing are also making it easier to design and produce complex microfluidic devices بسرعة and cost-effectively.
Wearable microfluidic devices are another exciting possibility. These systems could continuously monitor health parameters, providing real-time data and enabling early detection of medical conditions.
As these technologies mature, microfluidics has the potential to transform healthcare from a reactive system to a proactive one, where diseases are detected and treated before they become severe.
Conclusion
Microfluidics represents a paradigm shift in medicine, moving from large, centralized laboratories to compact, decentralized systems that bring diagnostic and analytical capabilities closer to patients. By leveraging principles of chemical engineering, materials science, and biology, lab-on-a-chip technology enables faster, more efficient, and more accessible healthcare solutions.
While challenges remain, the progress made so far suggests that microfluidics will play an increasingly important role in the future of medicine. From rapid diagnostics and personalized treatments to advanced drug development and organ-on-a-chip systems, the impact of this technology is far-reaching.
Ultimately, the lab-on-a-chip revolution is not just about miniaturizing laboratory processes—it is about redefining how healthcare is delivered, making it more responsive, precise, and inclusive for people around the world.