In the modern technological era, batteries are the backbone of innovation. From smartphones and laptops to electric vehicles (EVs) and renewable energy storage, the demand for efficient, safe, and high-capacity energy storage systems has never been greater. For the past three decades, lithium-ion batteries have been the dominant technology, enabling the portable electronics revolution and accelerating the transition to electric mobility. However, a new and promising competitor—solid-state batteries—is emerging, sparking what many call the “battery wars.”
This competition is not merely about which battery lasts longer or charges faster. At its core, it is a battle of chemical engineering principles, involving ion transport, thermodynamics, reaction kinetics, and materials design. Understanding these underlying factors reveals why lithium-ion batteries have succeeded, what limits them, and how solid-state batteries could reshape the future.
Fundamentals of Battery Chemistry
All batteries operate based on electrochemical reactions that convert chemical energy into electrical energy. They consist of three essential components: an anode (negative electrode), a cathode (positive electrode), and an electrolyte that facilitates ion movement between them.
E=E∘−RTnFlnQE = E^\circ – \frac{RT}{nF} \ln QE=E∘−nFRTlnQ
This thermodynamic relationship shows that the voltage of a battery depends on the chemical potentials of the reacting species. In practical terms, it means that battery performance is governed by both the materials used and the conditions inside the cell.
During discharge, oxidation occurs at the anode, releasing electrons that travel through an external circuit, while reduction occurs at the cathode. Simultaneously, ions move through the electrolyte to maintain charge balance. The efficiency of these processes determines how well a battery performs.
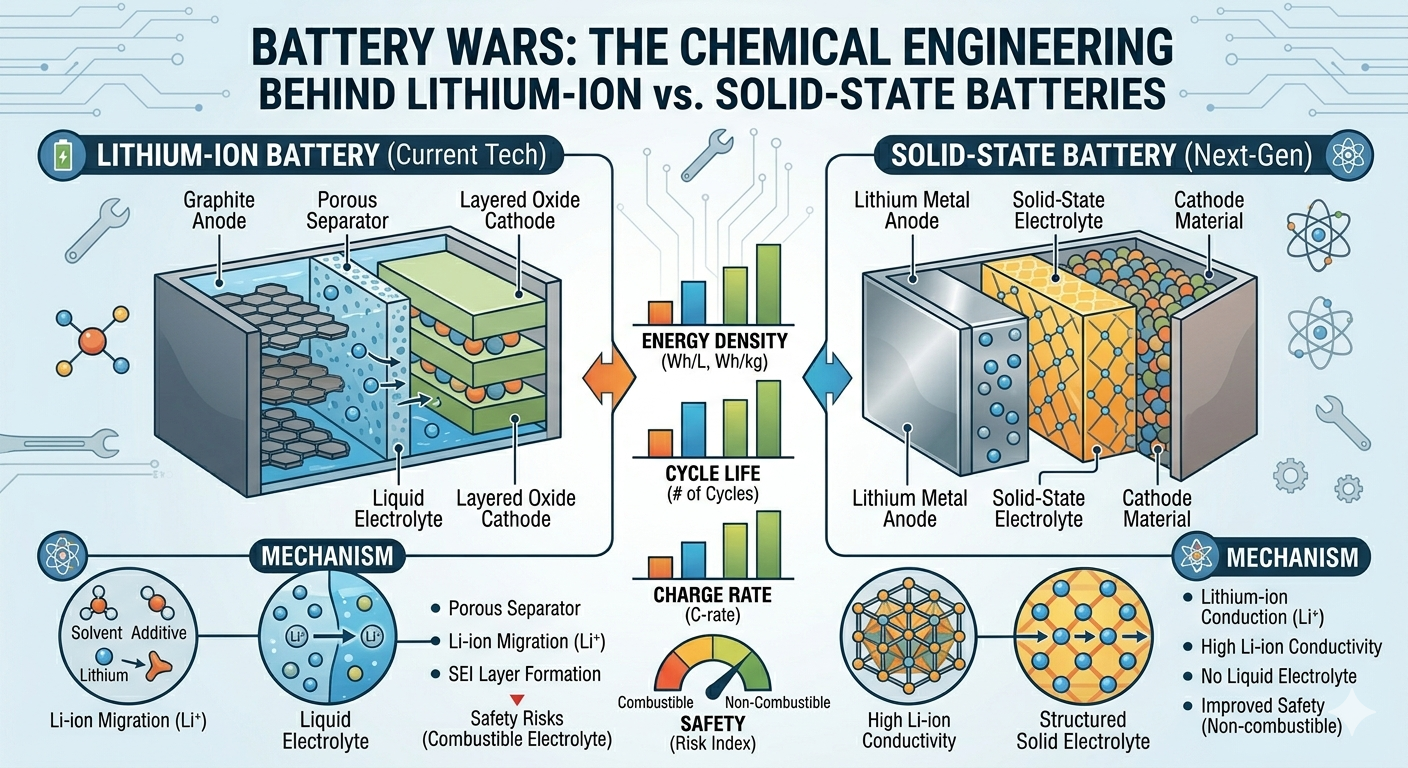
Lithium-Ion Batteries: The Established Standard
Lithium-ion batteries have become the industry standard due to their high energy density, long cycle life, and relatively mature manufacturing processes.
Structure and Operation
A typical lithium-ion battery includes:
- A graphite anode
- A lithium metal oxide cathode (such as lithium cobalt oxide or nickel manganese cobalt)
- A liquid electrolyte containing lithium salts
- A porous separator to prevent short circuits
During operation, lithium ions shuttle between the electrodes, intercalating into the electrode materials without causing major structural changes.
Advantages Rooted in Chemical Engineering
One of the biggest strengths of lithium-ion batteries is their high ionic conductivity. The liquid electrolyte allows lithium ions to move quickly, enabling fast charging and high power output. This property is critical for applications such as electric vehicles, where rapid energy delivery is essential.
Another advantage is the maturity of manufacturing processes. Chemical engineers have spent decades optimizing slurry preparation, electrode coating, drying, and cell assembly. These processes are now highly scalable, making lithium-ion batteries relatively cost-effective.
Lithium-ion systems also benefit from flexible chemistry. Engineers can modify cathode compositions, electrolyte formulations, and additives to balance performance, cost, and safety. For example, lithium iron phosphate (LFP) offers better thermal stability, while nickel-rich cathodes provide higher energy density.
Limitations and Challenges
Despite their success, lithium-ion batteries have inherent limitations.
One major concern is safety. The liquid electrolytes used are flammable and can lead to thermal runaway under extreme conditions, such as overcharging or physical damage. This has led to well-publicized battery fires in consumer electronics and electric vehicles.
Another limitation is energy density. Graphite anodes can only store a limited amount of lithium, placing a ceiling on how much energy can be packed into a given volume or weight.
Degradation is also a significant issue. Over time, lithium-ion batteries lose capacity due to processes such as:
- Formation of the solid electrolyte interphase (SEI)
- Electrolyte decomposition
- Lithium plating during fast charging
These mechanisms reduce battery lifespan and performance.
Solid-State Batteries: The Next Frontier
Solid-state batteries represent a fundamentally different approach. Instead of using a liquid electrolyte, they employ a solid electrolyte, which can be ceramic, polymer-based, or sulfide-based.
Working Principle
In solid-state batteries, lithium ions move through a solid material rather than a liquid. This eliminates the need for a separator and allows for new electrode configurations, including the use of lithium metal as the anode.
While the basic electrochemical principles remain the same, the shift from liquid to solid introduces significant changes in how ions move and how interfaces behave.
Advantages from a Chemical Engineering Perspective
One of the most compelling benefits of solid-state batteries is enhanced safety. Solid electrolytes are typically non-flammable and more resistant to high temperatures, significantly reducing the risk of fire.
Another major advantage is higher energy density. By enabling the use of lithium metal anodes, solid-state batteries can store much more energy than conventional lithium-ion systems. This could potentially double the range of electric vehicles or extend the battery life of portable devices.
Solid-state systems also show promise in reducing unwanted side reactions. The stable nature of solid electrolytes can limit degradation pathways, potentially improving cycle life.
Engineering Challenges
Despite their advantages, solid-state batteries face several critical challenges.
The most significant issue is ionic conductivity. While some solid electrolytes have achieved conductivities comparable to liquids, many still lag behind, limiting performance, especially at room temperature.
Another major challenge is interface resistance. In lithium-ion batteries, the liquid electrolyte naturally wets the electrode surfaces, ensuring good contact. In contrast, solid-state batteries rely on solid-solid interfaces, which can suffer from poor contact and high resistance.
Mechanical issues also arise due to volume changes during charging and discharging. These changes can lead to cracks or loss of contact between components, degrading performance over time.
Finally, manufacturing complexity remains a barrier. Producing solid-state batteries often requires high pressures, precise material control, and entirely new fabrication techniques, making them expensive and difficult to scale.
Comparing the Two Technologies
When comparing lithium-ion and solid-state batteries, it becomes clear that each has its strengths and weaknesses.
Lithium-ion batteries excel in:
- Established manufacturing
- High power output
- Lower cost
Solid-state batteries offer:
- Superior safety
- Higher potential energy density
- Improved thermal stability
However, solid-state technology is still in the development phase, while lithium-ion continues to evolve with incremental improvements.
The Role of Chemical Engineering
The competition between these two technologies is deeply rooted in chemical engineering disciplines.
Mass Transport
Efficient ion movement is critical. Engineers must design materials that allow fast diffusion while maintaining stability.
Thermodynamics
The choice of materials determines voltage, energy density, and stability limits.
Reaction Engineering
Understanding and controlling interfacial reactions is essential to prevent degradation.
Materials Science
Developing new electrolytes and electrode materials is at the heart of innovation.
Process Engineering
Scaling up production while maintaining quality and reducing cost is a major challenge, especially for solid-state batteries.
Real-World Applications and Impact
In electric vehicles, solid-state batteries could significantly increase driving range and improve safety. However, lithium-ion batteries remain dominant due to their reliability and cost advantages.
In consumer electronics, lithium-ion technology continues to meet current demands, though solid-state batteries may appear in premium devices in the future.
For grid storage, cost is the primary concern, making lithium-ion—particularly LFP chemistry—the preferred choice for now.
The Road Ahead
The future of energy storage is unlikely to be dominated by a single technology. Instead, we are likely to see a diverse ecosystem where different battery types serve different applications.
Lithium-ion batteries will continue to improve through innovations such as silicon anodes and advanced electrolytes. Meanwhile, solid-state batteries will gradually overcome their challenges and find their place in high-performance applications.
Conclusion
The “battery wars” between lithium-ion and solid-state technologies highlight the importance of chemical engineering in shaping the future of energy storage. While lithium-ion batteries remain the current champion due to their maturity and scalability, solid-state batteries offer a glimpse into a safer and more energy-dense future.
Ultimately, the outcome of this competition will depend not only on scientific breakthroughs but also on the ability to solve complex engineering challenges at scale. As research continues and technologies evolve, both battery types are likely to coexist, each playing a crucial role in powering the world of tomorrow.