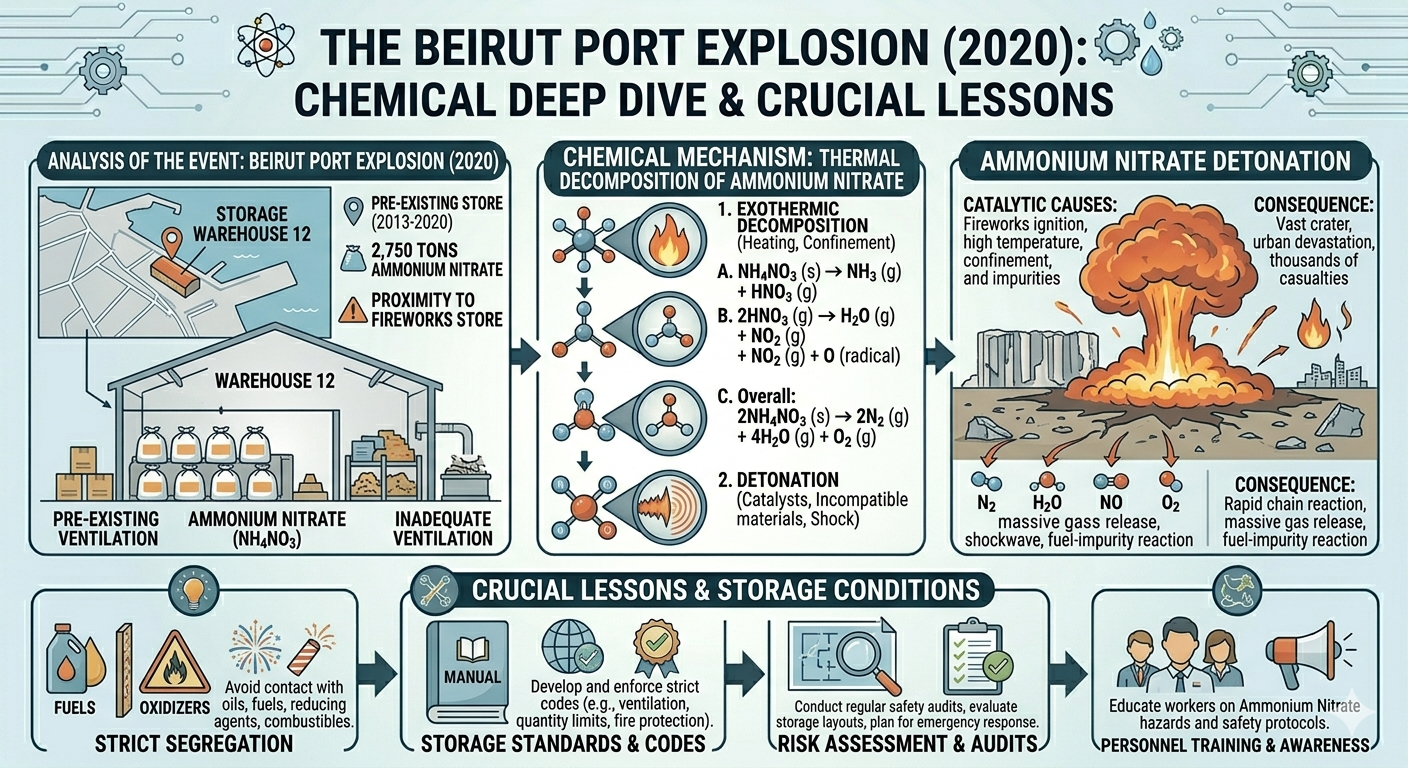
On August 4, 2020, a devastating explosion ripped through the port of Beirut, Lebanon, killing over 200 people, injuring thousands, and leaving large parts of the city in ruins. The disaster, now known as the Beirut Port Explosion, was caused by the detonation of approximately 2,750 tonnes of ammonium nitrate that had been stored improperly for years. While the scale of destruction shocked the world, the underlying cause was neither mysterious nor unprecedented. The hazards associated with ammonium nitrate are well understood in chemical engineering, and the explosion serves as a stark reminder that storage conditions are not optional—they are critical safety controls.
This article explores the chemistry of ammonium nitrate, the mechanisms of its thermal decomposition, and the engineering principles that explain why improper storage can turn a common industrial chemical into a catastrophic hazard.
Ammonium Nitrate: Useful but Potentially Dangerous
Ammonium nitrate (NH₄NO₃) is a widely used chemical with dual applications. In agriculture, it serves as an effective nitrogen-rich fertilizer, supporting crop growth across the globe. In industry, it functions as a powerful oxidizer in explosives such as ANFO (ammonium nitrate fuel oil), commonly used in mining and construction.
Under normal conditions, ammonium nitrate is considered stable and relatively safe. However, this stability is conditional, meaning it depends heavily on environmental factors such as temperature, contamination, confinement, and storage duration. When these conditions are not properly controlled, the compound can undergo rapid and violent decomposition.
The Chemistry of Thermal Decomposition
The danger of ammonium nitrate lies in its ability to decompose exothermically when exposed to heat. The simplest decomposition pathway is relatively mild:
NH4NO3→N2O+2H2O
In this reaction, ammonium nitrate breaks down into nitrous oxide (N₂O) and water vapor. Under controlled conditions, such as in industrial processes, this reaction can be managed safely.
However, under elevated temperatures or in the presence of contaminants, alternative and more dangerous pathways can occur. These reactions produce gases such as nitrogen, oxygen, and steam in large volumes, releasing significant energy in a very short time. The rapid expansion of gases creates high pressure, which can lead to an explosion if the system is confined.
This phenomenon is an example of thermal runaway, where the heat generated by a reaction accelerates the reaction itself. Once initiated, thermal runaway can become uncontrollable, resulting in catastrophic failure.
Why Ammonium Nitrate Becomes Explosive
Several factors contribute to the transformation of ammonium nitrate from a stable compound into an explosive hazard.
Oxidizing Nature
Ammonium nitrate is a strong oxidizer, meaning it supplies oxygen to support combustion. While it does not burn on its own, it can intensify fires and react violently when combined with combustible materials. If mixed with fuels such as oil, dust, or organic matter, it can form explosive mixtures.
Sensitivity to Contamination
Contaminants play a critical role in altering the behavior of ammonium nitrate. Substances such as hydrocarbons, metals, or even certain impurities can lower the activation energy required for decomposition, making the reaction more likely to occur and more violent when it does.
Effect of Confinement
In an open environment, gases produced during decomposition can escape, reducing pressure buildup. However, when ammonium nitrate is stored in enclosed or poorly ventilated spaces, pressure can accumulate rapidly. This confinement increases the likelihood of an explosion, as the expanding gases have nowhere to dissipate.
Exposure to Heat and Fire
External heat sources, such as fires, can initiate decomposition. Once the temperature exceeds a critical threshold, the reaction can accelerate rapidly, especially if heat is not removed efficiently.
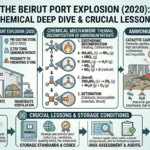
The Beirut Disaster: A Failure of Storage and Oversight
In the case of the Beirut Port Explosion, the ammonium nitrate had been stored in a warehouse for several years without proper safety measures. The material was reportedly kept in bulk, in proximity to other potentially hazardous substances, and without adequate ventilation or monitoring.
A fire in the warehouse is believed to have triggered the decomposition process. With large quantities of ammonium nitrate present and confined within the structure, the conditions were ideal for a runaway reaction. The result was a massive explosion equivalent to several hundred tonnes of TNT.
The disaster was not caused by a single failure but by a combination of factors, including negligence, poor risk management, and lack of regulatory enforcement.
Chemical Engineering Analysis
From a chemical engineering perspective, the Beirut explosion can be understood through several fundamental principles.
Heat Transfer
Effective heat removal is essential to prevent thermal runaway. In this case, the heat generated by the initial fire and subsequent decomposition was not dissipated, allowing temperatures to rise uncontrollably.
Reaction Kinetics
The rate of ammonium nitrate decomposition increases exponentially with temperature. Even a small increase in temperature can significantly accelerate the reaction, leading to rapid energy release.
Mass and Energy Balance
The decomposition process generates large volumes of gas and heat. If the system cannot accommodate this increase, pressure builds up, leading to mechanical failure and explosion.
Process Safety Management
Proper hazard identification and risk assessment could have identified the dangers associated with long-term storage of large quantities of ammonium nitrate. Techniques such as Hazard and Operability Studies (HAZOP) are designed to prevent such scenarios.
Why Storage Conditions Are Non-Negotiable
The safe handling of ammonium nitrate depends on strict adherence to storage guidelines. These conditions are not recommendations—they are essential controls.
Temperature Control
Ammonium nitrate must be stored away from heat sources and in environments where temperature can be monitored and controlled. Preventing overheating is critical to avoiding decomposition.
Isolation from Contaminants
The material should be kept separate from combustible substances, chemicals, and impurities. Even small amounts of contamination can significantly increase risk.
Proper Ventilation
Adequate ventilation allows heat and gases to dissipate, reducing the likelihood of pressure buildup and explosion.
Limiting Storage Quantity and Duration
Large stockpiles increase the potential impact of an incident. Minimizing the quantity stored and avoiding long-term storage reduces overall risk.
Fire Prevention and Protection
Facilities must implement fire detection and suppression systems, as well as strict controls to eliminate ignition sources.
Lessons Learned
The Beirut explosion highlights several critical lessons for industry and regulators alike.
First, known hazards must be taken seriously. The risks associated with ammonium nitrate are well documented, and failure to act on this knowledge can have devastating consequences.
Second, safety systems must be enforced and maintained. Regulations are only effective if they are implemented and monitored consistently.
Third, time increases risk. Prolonged storage of hazardous materials without proper oversight creates opportunities for failure.
Finally, small triggers can lead to large disasters. A relatively minor fire can escalate into a catastrophic event when hazardous materials are involved.
Broader Implications for Industry
The Beirut disaster is not an isolated incident. Similar events, such as the Texas City disaster of 1947, demonstrate recurring patterns of poor storage practices and inadequate safety management. These incidents underscore the need for a strong safety culture, where risks are continuously assessed and managed.
Industries handling hazardous chemicals must prioritize:
- Regular inspections and audits
- Clear accountability and responsibility
- Ongoing training and awareness
- Investment in safety infrastructure
Conclusion
The Beirut Port Explosion stands as a tragic example of what can happen when chemical safety principles are ignored. Ammonium nitrate, while useful and widely used, is inherently hazardous under certain conditions. Understanding its thermal decomposition and the factors that influence it is essential for safe handling.
From a chemical engineering standpoint, the disaster illustrates the importance of heat management, reaction control, and proper storage. More importantly, it reinforces a fundamental truth:
Chemicals behave according to the laws of science, not human assumptions.
When those laws are ignored, the consequences can be catastrophic. By respecting these principles and implementing strict safety measures, such tragedies can be prevented, ensuring that industrial progress does not come at the cost of human lives.