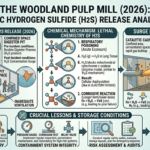
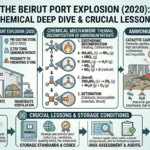
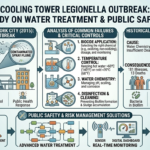
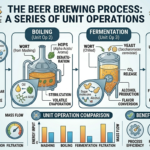
The strength of sulfuric acid is typically expressed in terms of its concentration or purity. The most common way to represent the strength of sulfuric acid is by indicating its molarity or the percentage of sulfuric acid in a solution. Here’s how to find the strength of sulfuric acid:
1. Molarity (Molar Concentration):
Molarity (M) is a measure of the number of moles of sulfuric acid (H2SO4) dissolved in one liter of solution. To find the molarity of sulfuric acid, you need to know:
- The mass of sulfuric acid (in grams) or the volume (in milliliters) of concentrated sulfuric acid.
- The molecular weight of sulfuric acid, which is 98.08 g/mol (2 hydrogen atoms, 1 sulfur atom, and 4 oxygen atoms).
The formula for calculating molarity is:
Molarity (M) = (Number of moles of solute) / (Volume of solution in liters)
Let’s say you have 50 grams of concentrated sulfuric acid (H2SO4). First, calculate the number of moles:
Number of moles of H2SO4 = (Mass of H2SO4) / (Molecular weight of H2SO4) Number of moles = 50 g / 98.08 g/mol ≈ 0.51 moles
Now, if you dissolve this 0.51 moles of sulfuric acid in 1 liter (1000 mL) of water, the molarity of the solution would be approximately 0.51 M.
2. Percentage Strength:
Sulfuric acid can also be expressed as a percentage strength, which represents the mass of sulfuric acid per 100 mL of the solution. To find the percentage strength, you need to know:
- The mass of sulfuric acid (in grams) present in the solution.
- The volume of the solution (in milliliters).
The formula for calculating the percentage strength is:
Percentage Strength (%) = (Mass of H2SO4 in solution / Volume of solution) x 100
For example, if you have 20 grams of sulfuric acid dissolved in 100 mL of water, the percentage strength would be:
Percentage Strength = (20 g / 100 mL) x 100% = 20%
So, the solution contains 20% sulfuric acid.
Please note that sulfuric acid can vary in strength, and it’s essential to handle concentrated sulfuric acid with extreme caution due to its corrosive nature. Always wear appropriate safety gear and follow safety protocols when working with strong acids. Additionally, check the label or product specifications for the precise strength of the sulfuric acid you are using, as commercial grades can differ in concentration.