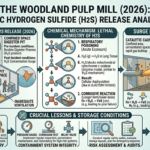
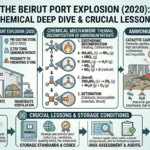
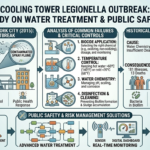
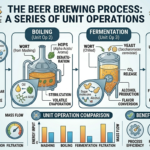
Introduction:
Entropy is a fundamental concept in physics, thermodynamics, information theory, and various other scientific disciplines. It is a measure of disorder or randomness in a system and has profound implications for our understanding of the physical world, the flow of energy, and the transmission of information. First introduced in the context of thermodynamics, entropy has since found applications in diverse fields, providing valuable insights into the behavior of natural systems. In this article, we delve into the concept of entropy, its significance, and its far-reaching implications across scientific domains.
What is Entropy?
In its simplest sense, entropy is a measure of the amount of disorder or randomness in a system. It is often associated with the idea that natural processes tend to move from a state of order to a state of disorder over time. The concept of entropy originated in the field of thermodynamics during the 19th century when scientists sought to understand the behavior of heat energy and its transfer in various systems.
In thermodynamics, entropy is linked to the notion of the second law, which states that the total entropy of an isolated system always increases over time. This law implies that heat spontaneously flows from hotter objects to cooler objects, and energy becomes more dispersed or spread out. This increase in entropy is often equated with a decrease in the amount of usable energy available to do work.
Entropy in Thermodynamics:
In thermodynamics, entropy is represented as S and is defined for a given system in terms of the amount of heat (Q) that is transferred reversibly to the system from its surroundings, divided by the temperature (T) of the system:
S = Q / T
This expression indicates that the change in entropy is directly related to the heat transfer and the temperature at which the heat is added or removed from the system. When heat is added to a system at a constant temperature, the entropy increases, and vice versa.
Entropy and Information Theory:
In the mid-20th century, the concept of entropy found a new application in information theory, thanks to the pioneering work of Claude Shannon. In information theory, entropy is a measure of the uncertainty or randomness in a set of data or information. It quantifies the average amount of information needed to describe an event or a message within a given set of possible events.
For example, consider a coin toss, where the outcome can be either heads or tails. If the coin is fair (equally likely to land heads or tails), the entropy of this event is at its maximum, as there is significant uncertainty about the outcome before the toss. On the other hand, if the coin is biased and more likely to land on one side, the entropy is reduced because there is less uncertainty about the result.
Implications in Various Fields:
Thermodynamics: Entropy helps explain the direction of natural processes, such as heat flow, energy dissipation, and the irreversibility of certain phenomena. It plays a crucial role in our understanding of energy transformations and the efficiency of heat engines.
Information Theory: Entropy is used to measure the efficiency of data compression and transmission, as well as the security of encryption algorithms. Higher entropy implies higher information content or randomness.
Cosmology: Entropy is related to the arrow of time in cosmology, where the universe is seen to move from a state of lower entropy (more ordered) in the past to a state of higher entropy (more disordered) in the future, as predicted by the second law of thermodynamics.
Biology: Entropy is also used in biological systems to describe the disorder in cellular processes and the transfer of energy in living organisms.
Conclusion:
Entropy is a fundamental concept with wide-ranging applications across scientific disciplines. Whether explaining the behavior of energy in thermodynamics, quantifying the uncertainty of information in communication systems, or examining the evolution of the universe, entropy provides valuable insights into the underlying order and randomness in natural systems. As we continue to explore the complexities of the physical world and the flow of information, entropy will undoubtedly remain a central and intriguing concept in our scientific endeavors.