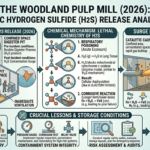
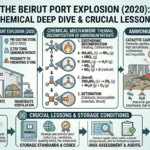
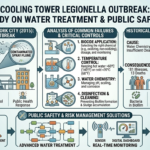
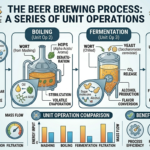
The global energy landscape is undergoing a monumental shift as nations commit to achieving net-zero greenhouse gas emissions by mid-century. This ambitious goal requires not only a massive expansion of renewable energy sources but also the development of new, sustainable fuels that can replace traditional fossil fuels across various sectors—including transportation, industry, and power generation. Among the promising candidates, e-methanol and e-ammonia have emerged as key players with the potential to revolutionize the way we produce, store, and use energy.
Produced from renewable electricity and abundant natural resources, e-methanol and e-ammonia offer carbon-neutral or carbon-free alternatives to fossil fuels. Their versatility, energy density, and compatibility with existing infrastructure make them attractive options for decarbonizing hard-to-electrify sectors such as shipping, heavy transport, and industrial processes. This article explores the science behind these fuels, their production pathways, advantages, challenges, and the future outlook for their widespread adoption.
Understanding E-Methanol and E-Ammonia
E-methanol and e-ammonia are synthetic fuels produced using renewable energy, typically via processes that involve electrolysis and chemical synthesis. The prefix “e” stands for “electro-” or “electrolytic,” highlighting the role of electricity in their manufacture.
E-Methanol is methanol (CH₃OH) synthesized by combining green hydrogen—produced by water electrolysis powered by renewable energy—with carbon dioxide (CO₂) captured from industrial emissions, biogenic sources, or even directly from the air. The result is a liquid fuel with many applications in transportation, power generation, and chemical manufacturing.
E-Ammonia is ammonia (NH₃) produced from nitrogen (N₂) in the air and green hydrogen via the Haber-Bosch process or emerging alternative methods. Ammonia is widely used as fertilizer today but is gaining attention as a carbon-free fuel option, especially for shipping and power generation.
Together, these fuels form part of the broader power-to-X (PtX) family, where surplus renewable electricity is converted into chemical energy carriers, enabling energy storage and flexible end-use.
Production Pathways
E-Methanol Production Process
The synthesis of e-methanol involves two critical steps:
Green Hydrogen Generation: Water (H₂O) is split into hydrogen (H₂) and oxygen (O₂) using electricity generated from renewable sources such as wind, solar, or hydropower. This process is known as electrolysis. Proton exchange membrane (PEM) and alkaline electrolyzers are the main technologies employed, with solid oxide electrolyzers emerging as a future option.
CO₂ Capture and Hydrogenation: Carbon dioxide is captured from industrial exhaust gases, biomass combustion, or directly from the atmosphere via direct air capture (DAC) technologies. The captured CO₂ is then reacted with green hydrogen over a catalyst—typically copper-based—under elevated temperature and pressure to form methanol and water:
CO2+3H2→CH3OH+H2O
Methanol produced this way is termed “e-methanol” and is considered carbon-neutral when the CO₂ feedstock is sourced sustainably.
E-Ammonia Production Process
The production of e-ammonia also relies on renewable hydrogen but combines it with nitrogen extracted from air:
Green Hydrogen Production: As with methanol, hydrogen is produced via renewable-powered water electrolysis.
Nitrogen Fixation: Nitrogen gas (N₂) from the air is combined with green hydrogen in the traditional Haber-Bosch process, which uses an iron-based catalyst at high temperature (~450°C) and pressure (~150-300 bar) to synthesize ammonia:
N2+3H2→2NH3
The energy-intensive Haber-Bosch process remains the dominant method for ammonia production globally. However, research is ongoing into electrochemical and plasma-based nitrogen fixation methods that could operate at lower temperatures and pressures, improving overall energy efficiency.
Advantages of E-Methanol and E-Ammonia
Carbon Neutrality and Decarbonization Potential
E-Methanol offers a circular carbon solution by recycling captured CO₂. When combusted, it releases the same amount of CO₂ that was used in its synthesis, resulting in net-zero emissions if renewable energy powers the entire process. It also serves as a building block for numerous chemicals and plastics, enabling sustainable feedstock substitution.
E-Ammonia is a carbon-free fuel; its combustion emits no CO₂, only nitrogen and water vapor. When produced using renewable hydrogen, it becomes a fully green fuel with enormous potential to decarbonize sectors where electrification is challenging.
Energy Density and Storage Advantages
E-Methanol is a liquid at ambient conditions with a high volumetric energy density (~4.4 kWh/L), making it easier to transport, store, and handle than gaseous fuels such as hydrogen. This facilitates the use of existing liquid fuel infrastructure with relatively minor adjustments.
E-Ammonia has a higher volumetric energy density (~3.2 kWh/L) than liquid hydrogen and can be stored as a liquid under moderate pressure or refrigeration. Its storability and transportability make it suitable for long-distance shipping and large-scale energy storage.
Versatility in Applications
E-Methanol can be used in internal combustion engines, fuel cells, and as a feedstock for synthetic hydrocarbons and plastics. Methanol-fueled engines have been tested in marine applications and are suitable for blending with gasoline.
E-Ammonia is emerging as a promising fuel for shipping, power plants, and industrial heating. It can be burned directly in thermal power plants or used in solid oxide fuel cells (SOFCs) for electricity generation without CO₂ emissions.
Infrastructure Compatibility
Methanol benefits from a mature supply chain and handling protocols due to its long-standing use in industry, enabling a relatively smooth transition.
Ammonia’s global production and distribution infrastructure, developed primarily for fertilizer use, can be adapted for fuel applications, reducing upfront investment costs.
Challenges to Overcome
Energy Intensity and Cost Barriers
Producing green hydrogen is still expensive compared to fossil-fuel-derived hydrogen, making e-methanol and e-ammonia costlier than conventional fuels. The overall efficiency of the production chain depends heavily on the availability of low-cost, abundant renewable electricity and improvements in electrolyzer technologies.
Safety and Handling Concerns
Methanol is toxic and flammable. Proper safety standards, spill management, and training are necessary to mitigate risks.
Ammonia is corrosive and toxic with a pungent smell. Leak detection and ventilation are critical, and safety measures must be stringent to prevent exposure and environmental harm.
Combustion and Emission Issues
Ammonia has a high ignition temperature and low flame speed, posing technical challenges for combustion in engines and turbines. Its combustion can generate nitrogen oxides (NOx), requiring effective emission control technologies.
Methanol combustion is cleaner than gasoline but can still produce trace formaldehyde and other pollutants that need to be managed.
Technological Development and Scaling
Scaling up electrolysis, CO₂ capture, and fuel synthesis technologies to commercial levels requires substantial investment and innovation. Additionally, alternative, more energy-efficient ammonia synthesis methods need to mature for broader adoption.
Applications and Market Potential
Marine and Shipping Sector
Maritime transport accounts for roughly 3% of global CO₂ emissions. Both e-methanol and e-ammonia are prime candidates for zero-emission marine fuels:
E-methanol is being trialed in methanol-powered ships, offering benefits like low sulfur oxide (SOx), particulate matter, and NOx emissions.
E-ammonia is viewed as a long-term solution for large vessels due to its carbon-free combustion and energy density, although technical challenges remain to optimize engines and fuel handling.
Power Generation and Grid Stability
Ammonia can be co-fired with traditional fuels or burned directly in thermal power plants and fuel cells, enabling renewable energy storage and grid balancing.
Methanol fuel cells provide clean, decentralized power generation for remote or off-grid locations.
Heavy-Duty and Road Transportation
Methanol blends can be used in internal combustion engines with minor adjustments, providing a transition fuel to hydrogen and electrification.
Research on ammonia-powered internal combustion engines and fuel cells for trucks and buses is ongoing, potentially enabling low-carbon heavy transport.
Future Outlook
The path to large-scale adoption of e-methanol and e-ammonia depends on continued technological advancements, cost reductions, and policy support:
Cost Reduction: Economies of scale and improvements in electrolyzer efficiency will lower green hydrogen costs, the key input for these fuels.
Integrated Production Facilities: Combining renewable power, electrolysis, carbon capture, and fuel synthesis into integrated plants will boost efficiency and economics.
Advanced Synthesis Technologies: Development of low-temperature ammonia synthesis and innovative methanol catalysts will enhance competitiveness.
Infrastructure and Safety Development: Upgrading fuel distribution networks and establishing rigorous safety standards are vital.
Policy and Incentives: Carbon pricing, subsidies, and international agreements will drive investment and market adoption.
Numerous pilot projects and industrial demonstrations worldwide highlight the growing momentum behind these fuels.
Conclusion
E-methanol and e-ammonia represent transformative solutions to the urgent challenge of decarbonizing global energy systems. Their production from renewable energy and abundant feedstocks, combined with versatile applications and compatibility with existing infrastructure, position them as cornerstone fuels in the transition to a sustainable, net-zero future.
Although technical, economic, and safety challenges remain, rapid innovation, falling renewable energy costs, and supportive policies are accelerating their development and deployment. As the world strives to meet climate targets and secure energy resilience, e-methanol and e-ammonia are poised to play pivotal roles in powering a cleaner, greener future.